Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


Multiplex Assay Kit for Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay)
IGHE; Immunoglobulin Heavy Constant Epsilon; Ig epsilon chain C region
(Note: Up to 8-plex in one testing reaction)
- Product No.LMA545Mu
- Organism SpeciesMus musculus (Mouse) Same name, Different species.
- Sample Typeserum, plasma, tissue homogenates, cell lysates, cell culture supernates and other biological fluids
- Test MethodDouble-antibody Sandwich
- Assay Length3.5h
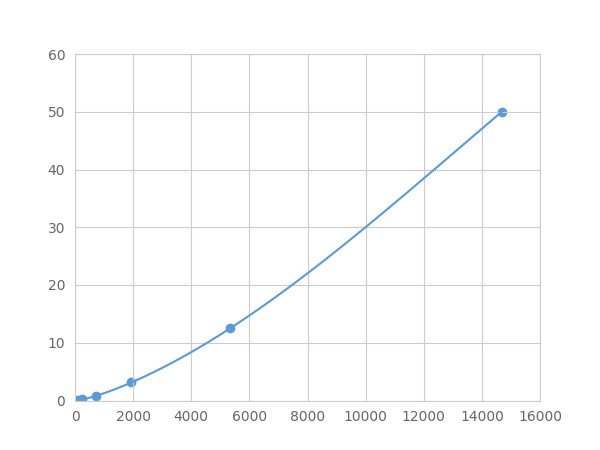
- Detection Range0.05-50ng/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 0.017 ng/mL.
- DownloadInstruction Manual
- UOM 8Plex 7Plex 6Plex 5Plex 4Plex 3Plex 2Plex1Plex
- FOB
US$ 405
US$ 420
US$ 443
US$ 475
US$ 506
US$ 552
US$ 622
US$ 778
Add to Price Calculator
Result
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay).
No significant cross-reactivity or interference between Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) and the recovery rates were calculated by comparing the measured value to the expected amount of Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 81-97 | 90 |
| EDTA plasma(n=5) | 85-94 | 91 |
| heparin plasma(n=5) | 84-99 | 90 |
| sodium citrate plasma(n=5) | 95-102 | 98 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 98-105% | 81-105% | 94-102% | 84-102% |
| EDTA plasma(n=5) | 78-97% | 85-103% | 93-101% | 85-104% |
| heparin plasma(n=5) | 80-99% | 79-104% | 95-102% | 94-105% |
| sodium citrate plasma(n=5) | 80-89% | 79-94% | 96-103% | 93-104% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| 96-well plate | 1 | Plate sealer for 96 wells | 4 |
| Pre-Mixed Standard | 2 | Standard Diluent | 1×20mL |
| Pre-Mixed Magnetic beads (22#:IgE) | 1 | Analysis buffer | 1×20mL |
| Pre-Mixed Detection Reagent A | 1×120μL | Assay Diluent A | 1×12mL |
| Detection Reagent B (PE-SA) | 1×120μL | Assay Diluent B | 1×12mL |
| Sheath Fluid | 1×10mL | Wash Buffer (30 × concentrate) | 1×20mL |
| Instruction manual | 1 |
Assay procedure summary
1. Preparation of standards, reagents and samples before the experiment;
2. Add 100μL standard or sample to each well,
add 10μL magnetic beads, and incubate 90min at 37°C on shaker;
3. Remove liquid on magnetic frame, add 100μL prepared Detection Reagent A. Incubate 60min at 37°C on shaker;
4. Wash plate on magnetic frame for three times;
5. Add 100μL prepared Detection Reagent B, and incubate 30 min at 37°C on shaker;
6. Wash plate on magnetic frame for three times;
7. Add 100μL sheath solution, swirl for 2 minutes, read on the machine.
GIVEAWAYS
INCREMENT SERVICES
| Magazine | Citations |
| Molecular Carcinogenesis | IL17A gene polymorphisms, serum IL-17A and IgE levels, and hepatocellular carcinoma risk in patients with chronic hepatitis B virus infection PubMed: 23280722 |
| Human Immunology | IL21 and IL21R polymorphisms and their interactive effects on serum IL-21 and IgE levels in patients with chronic hepatitis B virus infection PubMed: 23354321 |
| Scand J Immunol | Passive transfer of tumour-derived MDSCs inhibits asthma-related airway inflammation Pubmed: 24313384 |
| AAPS PharmSciTech | Topical Lyogel Containing Corticosteroid Decreases IgE Expression and Enhances the Therapeutic Efficacy Against Atopic Eczema PubMed: 25511806 |
| Ann Allergy Asthma Immunol | Reduced airway microbiota diversity is associated with elevated allergic respiratory inflammation PubMed: 26123423 |
| Molecular Medicine Reports | The protective role of vitamin D3 in a murine model of asthma via the suppression of TGF-β/Smad signaling and activation of the Nrf2/HO-1 pathway. pubmed:27484042 |
| Gut Pathog. | Oral administration of Clostridium butyricum CGMCC0313‐1 reduces ovalbumin‐induced allergic airway inflammation in mice pubmed:28250847 |
| International Immunopharmacology | Catalpol alleviates ovalbumin-induced asthma in mice: Reduced eosinophil infiltration in the lung pubmed:27992791 |
| Asian Pacific Society of Respirology | Oral administration of Clostridium butyricum CGMCC0313-1 reduces ovalbumin-induced allergic airway inflammation in mice pubmed:28122397 |
| Molecular Medicine Reports | Protective effects of astragaloside IV against ovalbumin-induced allergic rhinitis are mediated by T-box protein expressed in T cells/GATA-3 and forkhead box protein 3/retinoic acid-related orphan nuclear receptor γt. pubmed:28586019 |
| Journal of Autoimmunity | Commensal bacteria aggravate allergic asthma via NLRP3/IL-1β signaling in post-weaning mice 10.1016:j.jaut.2018.07.003 |
| Water Research | Sub-chronic inhalation of reclaimed water-induced fibrotic lesion in a mouse model Pubmed:29655095 |
| Human & Experimental Toxicology | Investigation of hypersensitivity potential of diacetyl by determining cytokine profiles Pubmed:29233007 |
| Journal of International Medical Research | Changes in Th1/Th2-producing cytokines during acute exacerbation chronic obstructive pulmonary disease Pubmed:29950127 |
| Journal Of Cellular Biochemistry | Effects of catalpol on bronchial asthma and its relationship with cytokines Pubmed: 30536454 |
| INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES | Adipose Tissue-Derived Mesenchymal Stem Cell Modulates the Immune Response of Allergic Rhinitis in a Rat Model Pubmed: 30781605 |
| Royal Society Open Science | Metabolomics analysis of baicalin on ovalbumin-sensitized allergic rhinitis rats |
| Health and Medicine | Antiallergical Effect of New Combined Nazal Aerodisperse System in the Conditions of Experimental Allergic Rhinitis |
| American Journal of Translational Research | Inhibitory effects of catalpol coordinated with budesonide and their relationship with cytokines and Interleukin-13 expression Pubmed: 31737193 |
| MOLECULAR IMMUNOLOGY | Animal models for analysis of hypersensitivity reactions to Shuanghuanglian injection Pubmed: 32302806 |
| bioscience reports | Protective effect of Asarum sieboldii essential oil on ovalbumin induced allergic rhinitis in rat Pubmed: 32395767 |
| international journal of antimicrobial agents | Synergistic Phage-surfactant Combination Clears IgE-promoted Staphylococcus aureus Aggregation in Vitro and Enhances the Effect in Vivo Pubmed: 32335278 |
| Aggravation of Airway Inflammation in RSV-Infected Asthmatic Mice Following Infection-Induced Alteration of Gut Microbiota | |
| ACS Biomaterials Science & Engineering | Tissue-Engineered Decellularized Allografts for Anterior Cruciate Ligament Reconstruction |
| Biomarkers of Allergic Asthma and their Association with Serum Parameters | |
| Biomed Pharmacother | Qingfei oral liquid inhibited autophagy to alleviate inflammation via mTOR signaling pathway in RSV-infected asthmatic mice 33706133 |
| Environ Pollut | Trace endotoxin in reclaimed water is only one of the risk sources in subchronic inhalation exposure 34090073 |
| Annals of Tropical Medicine & Public Health | Effectiveness of biomarkers and serum parameters in determination allergic asthma and detection of its severity |
| Mediators Inflamm | Roles Played by the PI3K/Akt/HIF-1α Pathway and IL-17A in the Chinese Subtype of Chronic Sinusitis with Nasal Polyps Pubmed:35075349 |
| Expert Opinion on Therapeutic Targets | Dapagliflozin mitigates ovalbumin-prompted airway inflammatory-oxidative successions and associated bronchospasm in a rat model of allergic asthma Pubmed:35549595 |
| Catalog No. | Related products for research use of Mus musculus (Mouse) Organism species | Applications (RESEARCH USE ONLY!) |
| NPA545Mu01 | Native Immunoglobulin E (IgE) | Positive Control; Immunogen; SDS-PAGE; WB. |
| EPA545Mu62 | Eukaryotic Immunoglobulin E (IgE) | Positive Control; Immunogen; SDS-PAGE; WB. |
| EPA545Mu63 | Eukaryotic Immunoglobulin E (IgE) | Positive Control; Immunogen; SDS-PAGE; WB. |
| EPA545Mu61 | Eukaryotic Immunoglobulin E (IgE) | Positive Control; Immunogen; SDS-PAGE; WB. |
| RPA545Mu01 | Recombinant Immunoglobulin E (IgE) | Positive Control; Immunogen; SDS-PAGE; WB. |
| RPA545Mu02 | Recombinant Immunoglobulin E (IgE) | Positive Control; Immunogen; SDS-PAGE; WB. |
| APA545Mu62 | Active Immunoglobulin E (IgE) | Cell culture; Activity Assays. |
| PAA545Mu01 | Polyclonal Antibody to Immunoglobulin E (IgE) | WB |
| PAA545Mu02 | Polyclonal Antibody to Immunoglobulin E (IgE) | WB; IHC; ICC; IP. |
| LAA545Mu71 | Biotin-Linked Polyclonal Antibody to Immunoglobulin E (IgE) | WB; IHC; ICC. |
| SEA545Mu | ELISA Kit for Immunoglobulin E (IgE) | Enzyme-linked immunosorbent assay for Antigen Detection. |
| SCA545Mu | CLIA Kit for Immunoglobulin E (IgE) | Chemiluminescent immunoassay for Antigen Detection. |
| LMA545Mu | Multiplex Assay Kit for Immunoglobulin E (IgE) ,etc. by FLIA (Flow Luminescence Immunoassay) | FLIA Kit for Antigen Detection. |