Packages (Simulation)

Reagent Preparation
Image (I)
Image (II)
Certificate


CLIA Kit for Platelet Factor 4 (PF4)
CXCL4; SCYB4; Chemokine C-X-C-Motif Ligand 4; Oncostatin-A; Iroplact
- Product No.SCA172Hu
- Organism SpeciesHomo sapiens (Human) Same name, Different species.
- Sample TypeSerum, platelet-poor plasma and cell culture supernates
- Test MethodDouble-antibody Sandwich
- Assay Length2h, 40min
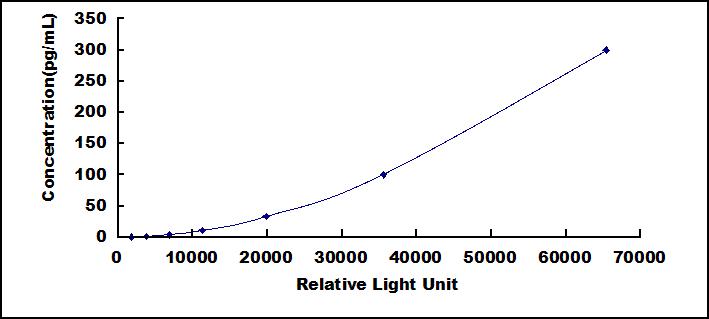
- Detection Range0.41-300pg/mL
- SensitivityThe minimum detectable dose of this kit is typically less than 0.17pg/mL.
- DownloadInstruction Manual
- UOM 48T96T 96T*5 96T*10 96T*100
- FOB
US$ 588
US$ 840
US$ 3780
US$ 7140
US$ 58800
For more details, please contact local distributors!
Specificity
This assay has high sensitivity and excellent specificity for detection of Platelet Factor 4 (PF4).
No significant cross-reactivity or interference between Platelet Factor 4 (PF4) and analogues was observed.
Recovery
Matrices listed below were spiked with certain level of recombinant Platelet Factor 4 (PF4) and the recovery rates were calculated by comparing the measured value to the expected amount of Platelet Factor 4 (PF4) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 84-103 | 98 |
| EDTA plasma(n=5) | 91-104 | 101 |
| heparin plasma(n=5) | 93-102 | 96 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Platelet Factor 4 (PF4) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Platelet Factor 4 (PF4) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Platelet Factor 4 (PF4) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 86-95% | 83-97% | 84-92% | 95-102% |
| EDTA plasma(n=5) | 89-96% | 89-96% | 95-104% | 86-104% |
| heparin plasma(n=5) | 85-98% | 89-96% | 90-98% | 94-103% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| Substrate A | 1×10mL | Substrate B | 1×2mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 100µL Substrate Solution. Incubate 10 minutes at 37°C;
8. Read RLU value immediately.
GIVEAWAYS
INCREMENT SERVICES
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 CLIA Kit Customized Service
CLIA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution
-
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
| Magazine | Citations |
| Biochimica et Biophysica Acta (BBA) - Proteins & Proteomics | Propofol lowers serum PF4 level and partially corrects hypercoagulopathy in endotoxemic rats PubMed: 20601223 |
| Basic Research in Cardiology | Secretome of apoptotic peripheral blood cells (APOSEC) attenuates microvascular obstruction in a porcine closed chest reperfused acute myocardial infarction model: role of platelet aggregation and vasodilation PubMed: 22899170 |
| PLoS One | Platelets Recognize Brain-Specific Glycolipid Structures, Respond to Neurovascular Damage and Promote Neuroinflammation PubMed: PMC3608633 |
| Diving and Hyperbaric Medicine | Effect of splenectomy on platelet activation and decompression sickness outcome in a rat model of decompression Pubmed:25311322 |
| Journal of Functional Foods | A Phellinus baumii–based supplement containing Salvia miltiorrhiza Bunge improves atherothrombotic profiles through endothelial nitric oxide synthase and cyclooxygenase pathwaysin vitro and in vivo science:S1756464616300676 |
| Plos one | Aspirin, but Not Tirofiban Displays Protective Effects in Endotoxin Induced Lung Injury pubmed:27583400 |
| international journal of molecular sciences | Evidence of the Role of R-Spondin 1 and Its Receptor Lgr4 in the Transmission of Mechanical Stimuli to Biological Signals for Bone Formation. pubmed:28272338 |
| Experimental and Therapeutic Medicine | Feasibility of improving platelet‑rich plasma therapy by using chitosan with high platelet activation ability pubmed:28450960 |
| Reproductive Toxicology | Aspirin pre-treatment modulates ozone-induced fetal growth restriction and alterations in uterine blood flow in rats Pubmed: 30528429 |
| INTERNATIONAL JOURNAL OF CARDIOLOGY | Short and long-term changes in platelet and inflammatory biomarkers after cryoballoon and radiofrequency ablation Pubmed: 30857843 |
| Available at SSRN 3335365 | Evaluating the Platelet Activation Related to the Degradation of Biomaterials by Scheme of Molecular Markers |
| journal of biotechnology | Newly Identified HNP‐F from Human Neutrophil Peptide 1 Promotes Hemostasis Pubmed: 30927490 |
| Data in Brief | Data for short and long-term prothrombotic biomarkers after cryoballoon and radiofrequency ablation |
| journal of thoracic and cardiovascular surgery | Evaluating Platelet Activation Related to the Degradation of Biomaterials Using Molecular Markers Pubmed: 32812629 |
| Marine Drugs | The Inhibitory Effect of Protamine on Platelets is Attenuated by Heparin without Inducing Thrombocytopenia in Rodents Pubmed: 31533230 |
| Thrombosis Research | Feasibility study of use of rabbit blood to evaluate platelet activation by medical devices Pubmed: 31838449 |
| Nat Commun | The C5a/C5a receptor 1 axis controls tissue neovascularization through CXCL4 release from platelets 34099640 |
| Vaccine | The effect of ChAdOx1 nCov-19 vaccine on arterial thrombosis development and platelet aggregation in female rats Pubmed:35183388 |