New findings on the regulation of T cell immunity
T cell lineage is an important immune cell population, which is an essential component of the adaptive immune system. The ligation of the T cell receptor (TCR) by antigen/MHC complexes and the interaction of costimulation receptors regulate T cell activation. After stimulation, naïve T cells begin to proliferate and differentiate into highly specific effector T cells (Teff). Diverse T cell subsets serve different but specific functions in response to a wide variety of stimuli. T cell immunity plays a critical role in host defenses to pathogens and immune responses against neoplasm. During alloimmunity, T cells play a pivotal role for both, in tolerance and rejection. Therefore, the study of T cell immune regulation mechanism is helpful for the treatment of pathogen infection, tumor, immune rejection and other diseases.
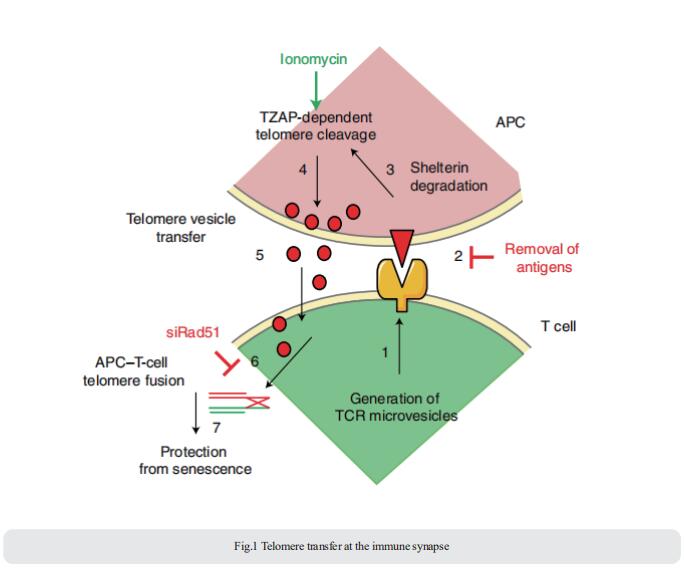
1. An intercellular transfer of telomeres rescues T cells from senescence and promotes long-term immunological memory
The common view is that T lymphocytes activate telomerase to delay senescence. Alessio Lanna, Department of Experimental and Translational Medicine, Division of Medicine, University College London, UK, and his team showed that some T cells (primarily naïve and central memory cells) elongated telomeres by acquiring telomere vesicles from antigen-presenting cells (APCs) independently of telomerase action[1]. Upon contact with these T cells, APCs degraded shelterin to donate telomeres, which were cleaved by the telomere trimming factor TZAP, and then transferred in extracellular vesicles at the immunological synapse (Fig.1). Telomere vesicles retained the Rad51 recombination factor that enabled telomere fusion with T-cell chromosome ends lengthening them by an average of ~3,000 base pairs. The telomere-acquiring T cells become stem-like and/or central long-lived memory cells, while other T cells commit to senescence. These telomere-acquiring T cells are protected from senescence before clonal division begins, conferring long-lasting immune protection.
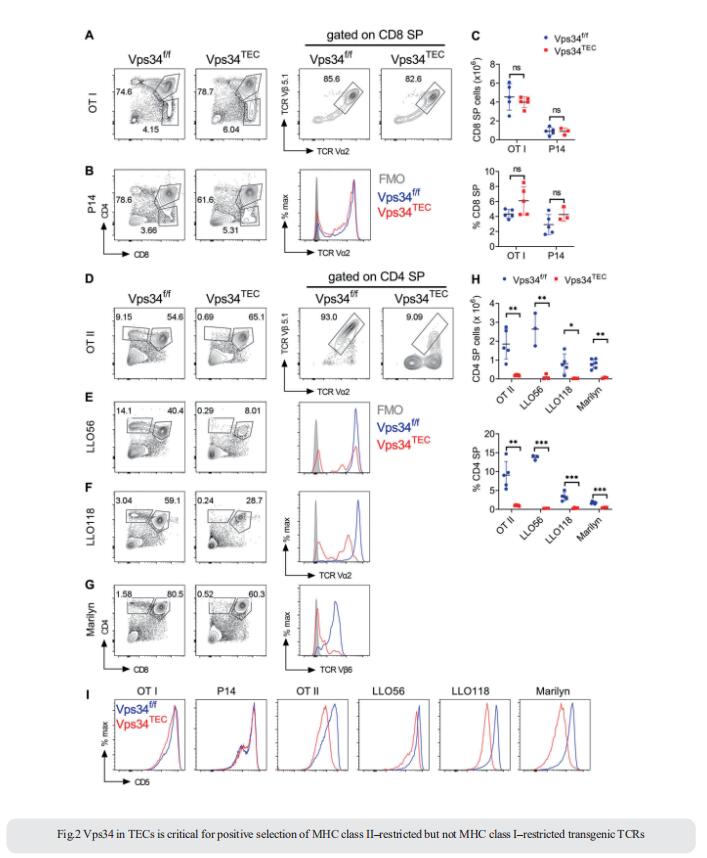
2. Thymic epithelial cells require lipid kinase Vps34 for CD4 but not CD8 T cell selection
The generation of a functional, self-tolerant T cell receptor (TCR) repertoire depends on interactions between developing thymocytes and antigen-presenting thymic epithelial cells (TECs). Cortical TECs (cTECs) rely on unique antigen-processing machinery to generate self-peptides specialized for T cell positive selection. Luc Van Kaer, Department of Pathology, Microbiology and Immunology, Vanderbilt University School of Medicine, USA, and his team showed that loss of Vps34 in TECs causes profound defects in the positive selection of the CD4 T cell lineage but not the CD8 T cell lineage[2]. Utilizing TCR sequencing, we show that T cell selection in conditional mutants causes altered repertoire properties including reduced clonal sharing. cTECs from mutant mice display an increased abundance of invariant chain intermediates bound to surface MHC class II molecules, indicating altered antigen processing (Fig.2). Collectively, these studies identified lipid kinase Vps34 as an important contributor to the repertoire of selecting ligands processed and presented by TECs to developing CD4 T cells.
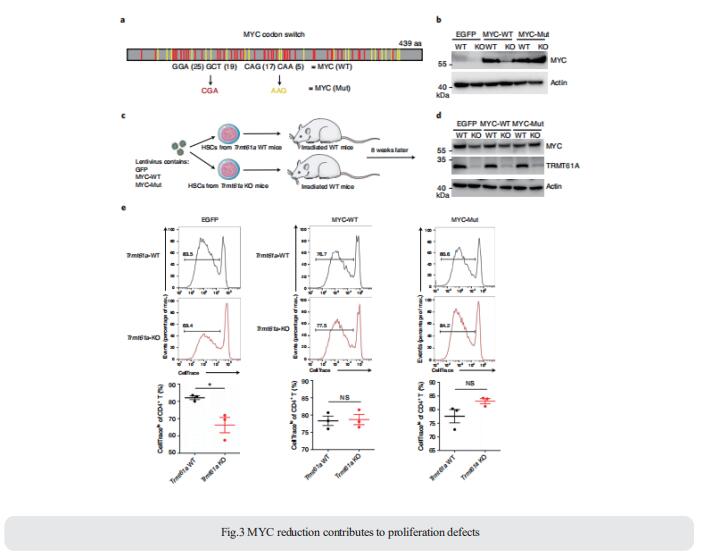
3. tRNA-m1A modification promotes T cell expansion via efficient MYC protein synthesis
Naive T cells undergo radical changes during the transition from dormant to hyperactive states upon activation, which necessitates de novo protein production via transcription and translation.However, the mechanism whereby T cells globally promote translation remains largely unknown. Hua-Bing Li, Shanghai Jiao Tong University School of Medicine, China, and his team showed that on exit from quiescence, T cells upregulate transfer RNA (tRNA) m1A58 ‘writer’ proteins TRMT61A and TRMT6, which confer m1A58 RNA modification on a specific subset of early expressed tRNAs[3]. These m1A-modified early tRNAs enhance translation efficiency, enabling rapid and necessary synthesis of MYC and of a specific group of key functional proteins. The MYC protein then guides the exit of naive T cells from a quiescent state into a proliferative state and promotes rapid T cell expansion after activation. Conditional deletion of the Trmt61a gene in mouse CD4+ T cells causes MYC protein deficiency and cell cycle arrest (Fig.3), disrupts T cell expansion upon cognate antigen stimulation and alleviates colitis in a mouse adoptive transfer colitis model. These results demonstrate that tRNA-m1A58 methylation serves as a translational checkpoint that constitutes an important mechanism responsible for the rapid synthesis of specific key functional proteins to promote T cell expansion.
References
[1]Lanna A, Vaz B, D'Ambra C, et al. An intercellular transfer of telomeres rescues T cells from senescence and promotes long-term immunological memory [J]. Nat Cell Biol. 2022, 10.1038/s41556-022-00991-z. (IF=28.213)
[2]Postoak JL, Song W, Yang G, et al. Thymic epithelial cells require lipid kinase Vps34 for CD4 but not CD8 T cell selection [J]. J Exp Med. 2022, 219(10):e20212554. (IF=17.579)
[3]Liu Y, Zhou J, Li X, et al. tRNA-m1A modification promotes T cell expansion via efficient MYC protein synthesis [J]. Nat Immunol. 2022, 10.1038/s41590-022-01301-3. (IF=31.250)
Cloud-Cloud can not only provide related products of T cell recognition, costimulation, proliferation, differentiation and effector molecules, such as CD3, CD4, CD8, TCR, MHC class Ⅰ/Ⅱ, CD28, CTLA4, IL-2, IL-4, IL-12, IFN-γ, etc. We also have the above-mentioned Rad51, Vps34, MYC and other related products, which can help the majority of scientific researchers to carry out T cell immune-related research.