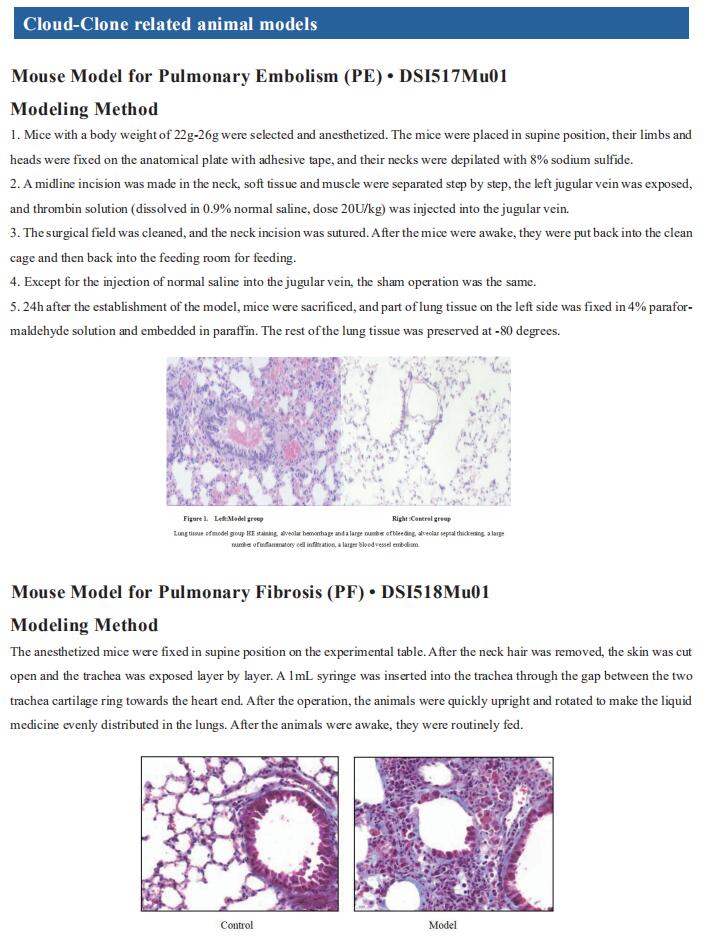
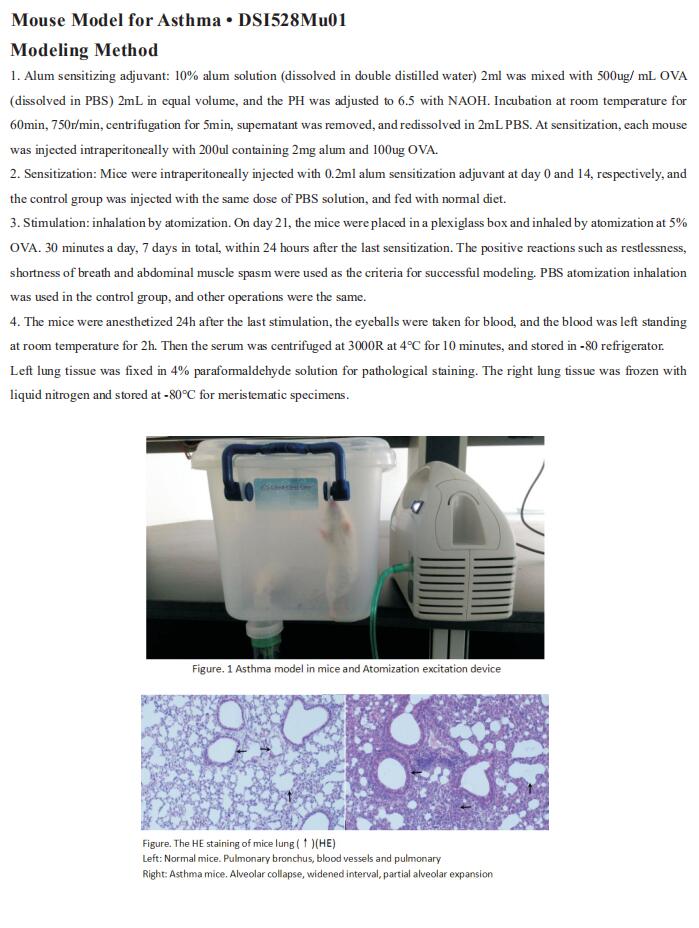
New findings on the mechanism of pulmonary arterial hypertension
Pulmonary arterial hypertension (PAH) is a fatal pulmonary vascular disease characterized by pulmonary vascular remodeling and increased pulmonary vascular resistance, culminating in right ventricular (RV) hypertrophy and failure. The dynamic vasoconstriction of pulmonary arteries, their adverse structural remodeling, fibrosis and stiffening, are the main causes of increased pulmonary vascular resistance. Besides this, endothelial dysfunction, an increase in inflammation, dysregulated angiogenesis, metabolic disturbance, oxidative stress and mitochondrial dysregulation contribute to the progression of the disease. Current treatments improve quality of life and survival primarily by dilating pulmonary arteries, but these agents do not address the mechanism underlying the progressive pulmonary vascular pathology. Therefore, it is necessary to further explore the mechanism of PAH occurrence and development to develop more effective therapeutic drugs
1. Endogenous Retroviral Elements Generate Pathologic Neutrophils in Pulmonary Arterial Hypertension
The role of neutrophils and their extracellular vesicles (EVs) in the pathogenesis of pulmonary arterial hypertension is unclear. Marlene Rabinovitch, Vera Moulton Wall Center for Pulmonary Vascular Diseases, Stanford University, USA, and his team related functional abnormalities in PAH neutrophils and their EVs to mechanisms uncovered by proteomic and transcriptomic profiling[1].
Neutrophils from PAH patients produce and release increased neutrophil elastase, associated with enhanced extracellular traps. They exhibit reduced migration and increased adhesion attributed to elevated β1 integrin and vinculin identified on proteomic analysis and previously linked to an antiviral response. This was substantiated by a transcriptomic interferon signature that they related to an increase in human endogenous retrovirus k envelope protein. Neutrophil EVs from patient plasma contain increased neutrophil elastase and human endogenous retrovirus k envelope and induce pulmonary hypertension in mice (Fig.1), mitigated by elafin, an elastase inhibitor. Taken together, the studies can explain how neutrophil dysfunction contributes to PAH and can be targeted therapeutically.
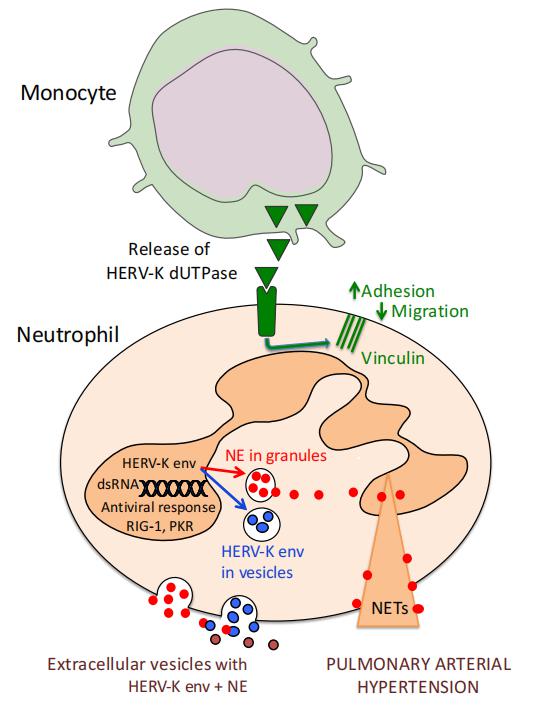
Fig.1 Elastase released from granules associates with HERV-K env in EVs causing pathologic features of PAH
2. HNRNPA2B1 Orchestrates Smooth Muscle Cell Phenotype in Pulmonary Arterial Hypertension
RNA-binding proteins are master orchestrators of gene expression regulation. In cancer, the RNA-binding protein HNRNPA2B1 (heterogeneous nuclear ribonucleoprotein A2B1; A2B1) promotes the pro-proliferative/anti-apoptotic phenotype. The same phenotype in pulmonary arterial smooth muscle cells (PASMCs) is responsible for the development of pulmonary arterial hypertension (PAH). Through the integration of computational and experimental biology, Grégoire Ruffenach, David Geffen School of Medicine, University of California, USA, and his team investigated the role of A2B1 in human PAH-PASMC[2]. They found that A2B1 expression and its nuclear localization are increased in human PAH-PASMC. In addition, they showed that in PAH-PASMC, A2B1 promotes the expression of its targets. A2B1 silencing in PAH-PASMC led to a decrease of all tested mRNAs carrying an A2B1 motif and a concomitant decrease in proliferation and resistance to apoptosis (Fig.2). In vivo A2B1 inhibition in the lungs rescued pulmonary hypertension in rats. The study revealed the role of A2B1 as a master orchestrator of the PAH-PASMC phenotype and its relevance as a therapeutic target in PAH.
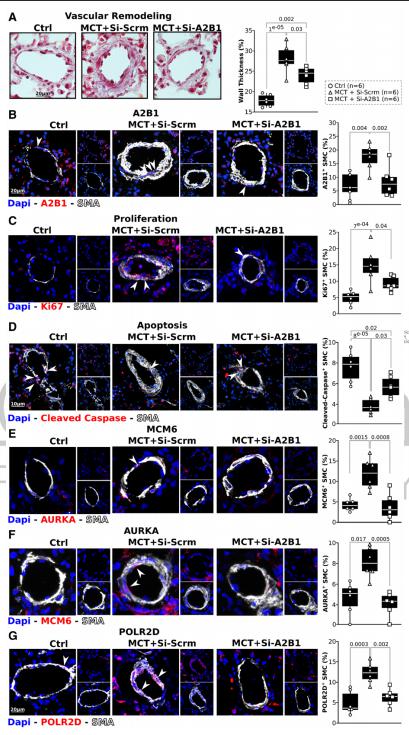
Fig.2 Pulmonary inhibition of A2B1 reverses vascular remodeling and the pro-proliferative/anti-apoptotic phenotype of SMCs
3. JAGGED-NOTCH3 signaling in vascular remodeling in pulmonary arterial hypertension
The NOTCH3 pathway is crucial in controlling vascular smooth muscle cell proliferation and maintaining smooth muscle cells in an undifferentiated state. PATRICIA A. THISTLETHWAITE, Division of Cardiothoracic Surgery, University of California, USA, and his team show that human PAH is characterized by overexpression of the NOTCH ligand JAGGED-1 (JAG-1) in small pulmonary artery smooth muscle cells and that JAG-1 selectively controls NOTCH3 signaling and cellular proliferation in an autocrine fashion[3]. In contrast, the NOTCH ligand DELTA-LIKE 4 is minimally expressed in small pulmonary artery smooth muscle cells from individuals with PAH, inhibits NOTCH3 cleavage and signaling, and retards vascular smooth muscle cell proliferation. Inhibition of JAG-1–induced NOTCH3 signaling in the lung reverses clinical and pathologic pulmonary hypertension in two rodent models of disease (Fig.3). The study proposed that selectively targeting JAG-1 activation of NOTCH3 may be an effective, safe strategy to treat PAH.
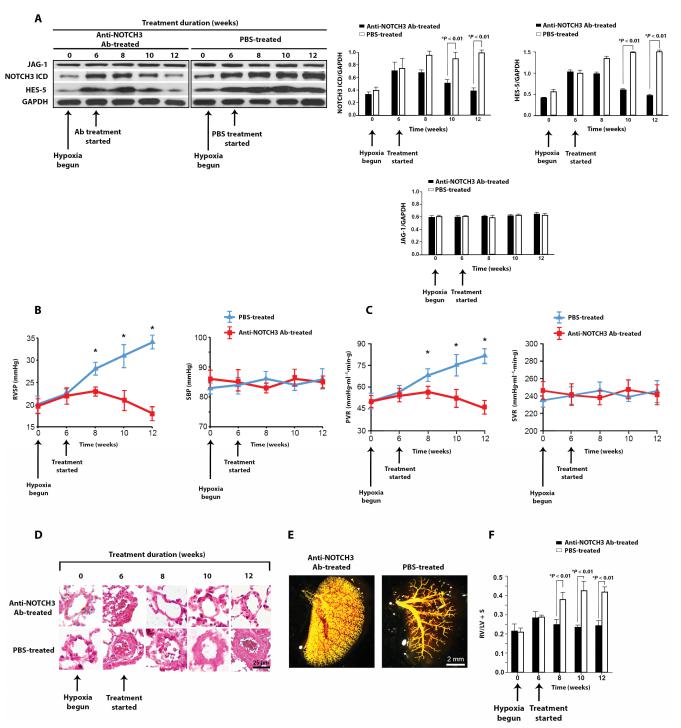
Fig.3 Anti-NOTCH3 Ab treatment reverses the development of hypoxic PAH in mice
4. Targeting peptidyl-prolyl isomerase 1 in experimental PAH
Peptidylprolyl cis/trans isomerase, NIMA interacting 1 (Pin1), a highly conserved enzyme, which binds to and catalyzes the isomerization of specific phosphorylated Ser/Thr-Pro motifs, acting as a molecular switch in multiple coordinated cellular processes. Tatyana Novoyatleva, Universities of Giessen and Marburg Lung Center (UGMLC), Germany, and his team investigated the expression and the role of Pin1 in experimental and clinical PAH by using Juglone, a specific and irreversible Pin1 inhibitor, established to covalently inactivate Pin1[4]. They demonstrated that the expression of Pin1 was markedly elevated in experimental PAH and PASMCs of patients with clinical PAH. In vitro Pin1 inhibition by either Juglone treatment or siRNA knock-down resulted in an induction of apoptosis and decrease in proliferation of human pulmonary vascular cells (Fig.4). Juglone administration lowered pulmonary vascular resistance, enhanced RV function, improved pulmonary vascular and cardiac remodeling in the Sugen/hypoxia rat model of PAH and the chronic hypoxia-induced PAH model in mice. The study demonstrates that targeting of Pin1 with small molecule inhibitor, Juglone, might be an attractive future therapeutic strategy for PAH and right heart disease secondary to PAH.
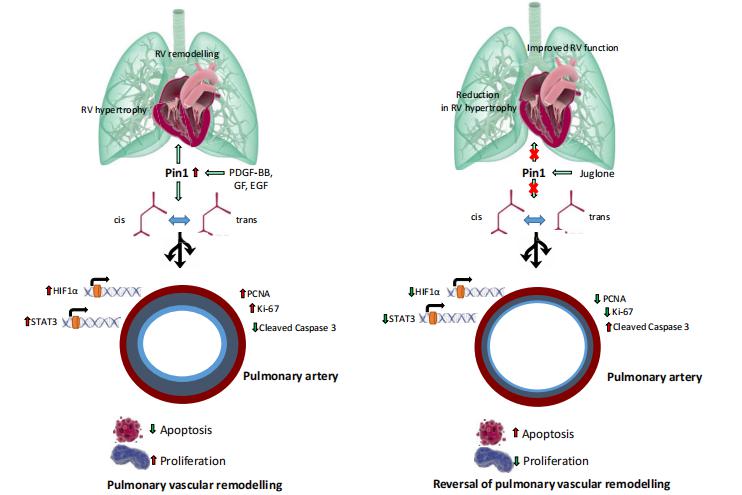
Fig.4 A proposed signaling mechanism of Pin1 in pulmonary hypertension
References
[1]Taylor S, Isobe S, Cao A, et al. Endogenous Retroviral Elements Generate Pathologic Neutrophils in Pulmonary Arterial Hypertension [J]. Am J Respir Crit Care Med. 2022, 10.1164/rccm.202102-0446OC. (IF=30.528)
[2]Ruffenach G, Medzikovic L, Aryan L, Li M, Eghbali M. HNRNPA2B1: RNA-Binding Protein That Orchestrates Smooth Muscle Cell Phenotype in Pulmonary Arterial Hypertension [J]. Circulation. 2022, 101161CIRCULATIONAHA122059591. (IF=39.918)
[3]Zhang Y, Hernandez M, Gower J, et al. JAGGED-NOTCH3 signaling in vascular remodeling in pulmonary arterial hypertension [J]. Sci Transl Med. 2022, 14(643):eabl5471. (IF=19.319)
[4]Rai N, Sydykov A, Kojonazarov B, et al. Targeting peptidyl-prolyl isomerase 1 in experimental pulmonary arterial hypertension [J]. Eur Respir J. 2022, 60(2):2101698. (IF=33.795)
Cloud-Clone can not only provide a variety of lung disease models, including pulmonary arterial hypertension, pulmonary edema, emphysema, chronic obstructive pulmonary disease, pulmonary fibrosis, pulmonary embolism, pneumonia, etc., covering common lung diseases. We also have various common detection indicators of lung diseases and the above HNRNPA2B1, JagGED-1, Delta-like 4, Pin1 and other related products, which can help the majority of scientific researchers to conduct research on lung diseases.