Focus on the IL6 Family: The Molecular Hub Connecting Immunity, Inflammation and Major Diseases
Focus on the IL6 Family: The Molecular Hub Connecting Immunity, Inflammation and Major Diseases
1. Introduction to the interleukin 6 family
The interleukin (IL) 6 family is a group of cytokines consisting of IL6, IL11, IL31, ciliary neurotrophic factor (CNTF), leukemia inhibitory factor (LIF), tumor necrosis factor M (OSM), cardiac trophic factor 1 (CT1), and cardioid cytokine (CLC). Members of this cytokine family have pro-inflammatory and anti-inflammatory properties, activating target genes involved in differentiation, survival, apoptosis and proliferation, and are major participants in hematopoiesis, acute phase and the body's immune response.
1.1 Structure of IL6 and Its Receptor
IL6 is a single-chain glycoprotein composed of four helical bundles. Helices A and B extend in one direction, while C and D extend in the opposite direction. IL6 transmits its signal through a cell surface type I receptor complex, which consists of a ligand-binding glycoprotein called IL6 receptor (IL6R) and a signal transduction component glycoprotein 130 (gp130). IL6R is an 80 kDa α chain and is also known as CD126. It consists of three domains, namely D1, D2, and D3. D1 is also known as the immunoglobulin (Ig) domain, and D2 and D3 constitute the extracellular cytokine binding domain (CBD). gp130 is composed of a β chain and is also known as CD130. gp130 consists of six domains (D1-D6). Similar to IL6R, D1 is the Ig domain, and D2 and D3 constitute the CBD. The other three domains (D4-D6) are important components for binding to IL6 and stimulating the subsequent signaling mechanism.
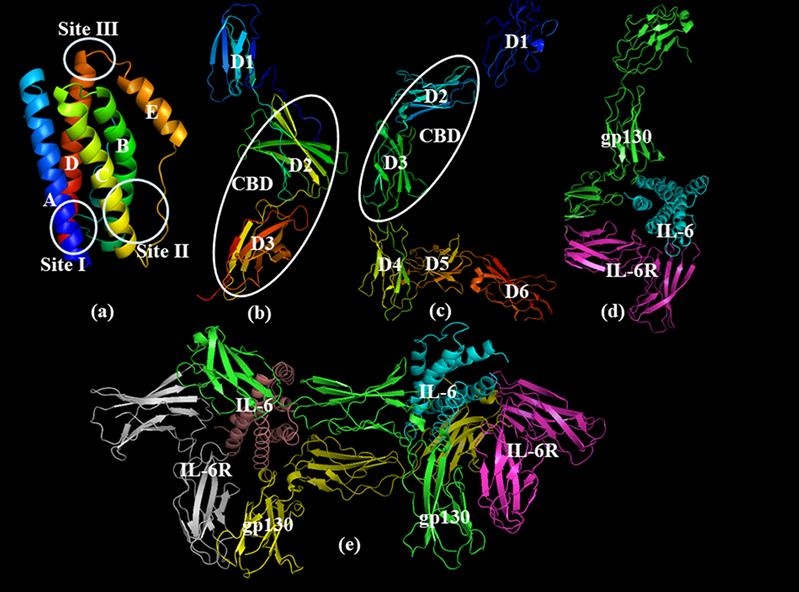
Fig. 1. Structure representation of IL6 and its receptor
(The figure is sourced from Bioorg Med Chem[1])
1.2 IL6 Signal Transduction Pathway
The signal transduction of IL6 can be divided into classical signal transduction and trans-signal transduction based on its binding receptors. IL6 binds to IL6R, which then associates with the signal-transducing receptor chain glycoprotein 130 (gp130), thereby activating the JAK/STAT and MAPK cascades. There are two types of IL6R. Membrane-bound IL6R is expressed only by a limited number of cells, including hepatocytes and certain lymphocyte subsets, and mediates classical signal transduction. Therefore, the classical signal transduction pathway mediated by IL6 is restricted to specific cell types. The other form of IL6R is soluble IL6R (sIL6R), which is expressed through proteolytic cleavage of the membrane IL6R or through alternative splicing of mRNA for translation, and it mediates trans-signal transduction. IL6 binds to sIL6R to form a complex, which subsequently activates intracellular signaling through gp130. Since nearly all cells express gp130, trans-signal transduction of IL6 is present in almost all cell types.
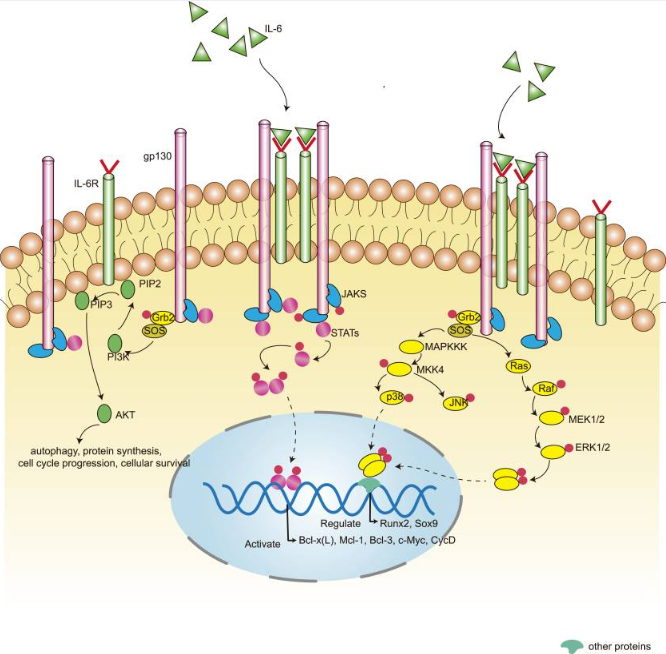
Fig.2 IL6 classical signal pathway
(The figure is sourced from Kidney Dis (Basel)[2])
1.3 Function of the IL6 Family
The IL6 family is a group of highly versatile and core signaling proteins. They activate signaling pathways such as JAK/STAT and MAPK by sharing the gp130 receptor, playing a "double-edged sword" role in the physiological and pathological processes of the body. On one hand, they dominate acute immune defense, promote tissue repair, regulate metabolism, and maintain homeostasis; on the other hand, their excessive or continuous activation can drive chronic inflammation, autoimmune diseases, fibrosis, and cancer progression. Therefore, this family is a key hub connecting immunity, metabolism, and tissue function, and is also an important target for disease treatment.
2. Research on the IL6 Family and Cancer
The IL6 family cytokines are regarded as key factors in the relationship between inflammation and cancer. The overexpression of IL6 is associated with tumor progression, and its mechanisms include inhibiting apoptosis of cancer cells, stimulating angiogenesis, and enhancing drug resistance[3-7]. By inhibiting the IL6/STAT3 pathway, such as using the IL6R inhibitor Tocilizumab, the promoting effect of double cortin-like kinase 1 (DCLK1) on the malignant phenotype of triple-negative breast cancer (TNBC) cells can be eliminated[8]. The IL6 neutralizing antibody Siltuximab may inhibit STAT3 tyrosine phosphorylation in a cell-dependent manner, thereby inhibiting the growth of lung cancer cells in vivo[9]. The selective estrogen receptor modulator Bazedoxifene may interfere with the protein interaction between IL6 family proteins and gp130, which limits the growth of intestinal tumors by inhibiting IL11-mediated signal transduction[10]. A small molecule compound, SMI-10B13, can inhibit the OSM signal. In a human breast cancer mouse model, SMI-10B13 reduces tumor growth and improves the survival rate of mice[11]. LIF is a key paracrine factor that activates pancreatic stellate cells to act on cancer cells. Drug-induced LIF blockade significantly slows tumor progression and enhances the efficacy of chemotherapy to prolong the survival period of mice with pancreatic ductal adenocarcinoma[12]. These studies highlight the potential clinical benefits of targeting IL6 family members in cancer treatment.
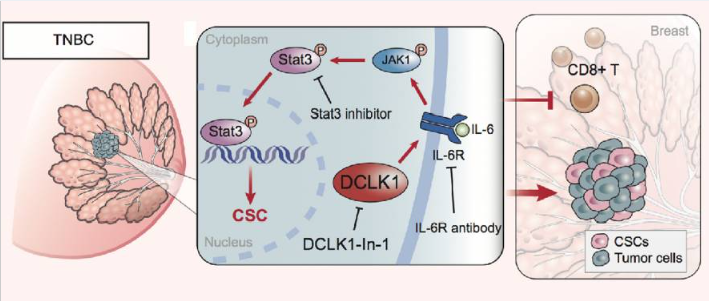
Fig.3 The IL6/STAT3 pathway activated by DCLK1 plays a crucial role in the malignant phenotype and anti-tumor immunity of TNBC
(The figure is sourced from Breast Cancer Res[8])
3. Research on the IL6 Family and Autoimmune diseases
The IL6 family proteins have been well described as the main factors of chronic inflammation, and chronic inflammation is crucial for the development of autoimmune diseases. Systemic lupus erythematosus (SLE) is an autoimmune disease caused by an immune system disorder. Intraperitoneal administration of anti-IL6 monoclonal antibodies inhibits the production of SLE-related autoantibodies and can prevent the development of severe kidney disease[13]. IL6 helps to induce skin sclerosis by bleomycin, and IL6 receptor-specific monoclonal antibodies may improve the symptoms of scleroderma by inhibiting fibroblast activation[14]. IL11 has a dual role in the pathogenesis of rheumatoid arthritis (RA), enhancing the infiltration of synovial fibroblasts and further aggravating the severity of the disease by increasing the invasion of blood vessels into RA pannus[15]. OSM is highly expressed in the kidney tissues of lupus nephritis (LN) mice. Anti-OSM antibodies can improve tubular epithelial-mesenchymal transition, inflammation, and renal tubular interstitial fibrosis, and partially improve 24-hour urine protein excretion and blood urea nitrogen production[16]. In herpes-type pemphigus (PH), the number of subcutaneous IL31 cells and IL31RA cells increases. The enhanced IL31/IL31RA signal and the increase in the number of skin eosinophils and basophils may be involved in the pruritus in PH[17]. Based on this, targeting the IL6 family pathway has become an important therapeutic strategy, significantly improving the clinical prognosis of autoimmune diseases.
4. Research on the IL6 Family and Cardiovascular diseases
Among inflammatory mediators, the IL6 family proteins are key factors in the pathophysiology of cardiovascular diseases. Multiple association analysis studies have shown that IL6 is significantly associated with major adverse cardiovascular events and the risks of cardiovascular death or heart failure[18-20]. After myocardial infarction, neutralizing the IL6 receptor inhibited myocardial inflammation, thereby alleviating left ventricular remodeling[21]. Clinical studies have shown that the IL6 inhibitor zavechimab significantly reduced the inflammatory and thrombosis biomarkers associated with atherosclerosis[22]. In advanced atherosclerosis models, inhibiting CT1 promoted anti-inflammatory and atherosclerotic protective effects, thereby eliminating the progression of atherosclerosis[23]. Elevated plasma CT1 levels are associated with the risk of heart failure in patients with hypertension and can serve as a biomarker for determining the prognosis of patients with hypertension[24]. In mice with Marfan syndrome (MFS), the IL11 mRNA and protein in the aorta increase, and inhibiting the IL11 pathway alleviated the disease symptoms of MFS mice[25]. OSM promotes the production of Notch3 by facilitating Notch3 expression, inhibits cardiomyocyte apoptosis, thereby activating the PI3K/Akt pathway, and thus alleviates cardiac ischemia/reperfusion (I/R) injury[26]. LIF can prevent the progression of pre-formed atherosclerotic plaques, affecting lesion size and vascular reactivity[27]. These results indicate that members of the IL6 family are potential intervention targets for the prevention and treatment of cardiovascular diseases.
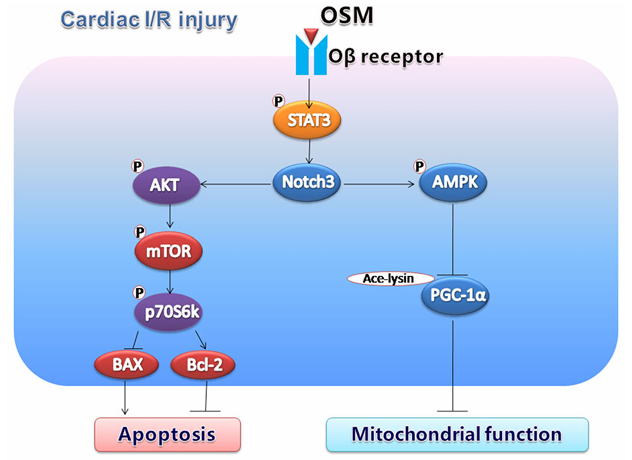
Fig.4 The possible mechanism of protective effect of OSM in cardiac I/R injury
(The figure is sourced from Apoptosis[26])
5. Research on the IL6 Family and Fibrotic diseases
Since inflammation is closely related to fibrosis, an increasing number of studies have shown that members of the IL6 family play an important role in organ fibrosis. In IL6 knockout (KO) mice, high-salt diet or angiotensin II-induced cardiac dysfunction and fibrosis were reduced[28]. Blocking IL6 signal transduction decreased the inflammatory level, immune cell infiltration, and expression of pro-fibrotic cytokines in renal tissue of the unilateral ureteral obstruction (UUO) mouse model, inhibiting the progression of renal fibrosis[29]. Trans-signaling of IL6 is a key driver of pulmonary allograft fibrosis[30]. In mice, specific IL11 transgenic expression or recombinant protein injection leads to cardiac and kidney fibrosis and organ failure[31]. Anti-IL11 treatment alleviated lung inflammation in bleomycin-induced pulmonary fibrosis mouse model and reversed pulmonary fibrosis[32]. OSM exerts strong fibrogenic activity during chronic liver injury by regulating macrophage activation[33]. In the mouse model of renal tubulointerstitial fibrosis (TIF), overexpression of LIF aggravates TIF[34]. Administration of LIF neutralizing antibodies to mice showed significant efficacy against TIF. Unlike other family members, exogenous CT1 administration can alleviate renal fibrosis in UUO mice by counteracting inflammation and apoptosis[35]. Therefore, targeting this family signaling pathway has become an important strategy for anti-fibrotic therapy.
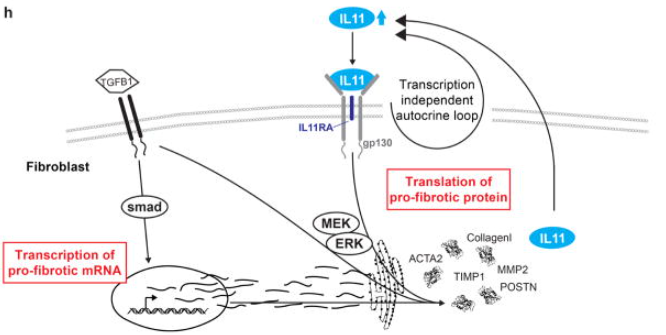
Fig.5 The fibrotic-promoting effect of IL11
(The figure is sourced from Nature[31])
Cloud-Clone supports scientific research and provides relevant detection reagent products for a wide range of scientific researchers. The core product numbers of the relevant targets are as follows:
Target | core product No. | Target | core product No. | Target | core product No. |
AKT1 | C231 | IL31RA | E763 | OSMR | B761 |
AKT2 | B719 | IL6 | A079 | PDK1 | C718 |
AKT3 | A382 | IL6R | B815 | PIK3Cb | J829 |
CLCF1 | C389 | IRS1 | C546 | PIK3Cd | J832 |
CNTF | A021 | IRS2 | D880 | PKCd | A433 |
CNTFR | C185 | JAK1 | C551 | PTPN11 | D584 |
CREB | B318 | JAK2 | F494 | Rac1 | M427 |
CRLF1 | F303 | JAK3 | F493 | Raf-1 | C232 |
CT1 | A810 | JNK1 | B156 | RASA1 | B616 |
ERK1 | B357 | JNK2 | D576 | RPS6Kb1 | L979 |
ERK2 | A930 | Jun | B292 | RPTOR | M681 |
FOS | B291 | JunB | H765 | SHC1 | E671 |
FOSL1 | J089 | LIF | A085 | SORT1 | C895 |
GAB2 | L533 | LIFR | A561 | STAT1 | B740 |
GbL | N253 | MAP2K1 | D559 | STAT2 | B796 |
gp130 | A046 | MAP2K2 | D562 | STAT3 | B743 |
GRB2 | C514 | MAP2K4 | MKK4 | STAT4 | B739 |
GSK3a | A630 | MAPK11 | B435 | STAT5A | B738 |
GSK3b | D317 | MAPK12 | D577 | STAT5B | B727 |
IL11 | A057 | MAPK13 | D578 | STAT6 | B737 |
IL11Ra | E771 | MAPK14 | B206 | TSC1 | C813 |
IL27A | A385 | MAPK7 | B431 | TYK2 | B595 |
IL27Ra | B194 | mTOR | B806 | Vav1 | C213 |
IL31 | B179 | OSM | A110 |
For more scientific research reagents, please visit the official website of Cloud-Clone:http://www.cloud-clone.com/
References
[1]Kaur S, Bansal Y, Kumar R, Bansal G. A panoramic review of IL-6: Structure, pathophysiological roles and inhibitors. Bioorg Med Chem. 2020;28(5):115327.
[2]Chen Y, Zhou J, Xu S, Nie J. Role of Interleukin-6 Family Cytokines in Organ Fibrosis. Kidney Dis (Basel). 2023;9(4):239-253.
[3]Ara T, Declerck YA. Interleukin-6 in bone metastasis and cancer progression. Eur J Cancer. 2010;46(7):1223-1231.
[4]Belluco C, Nitti D, Frantz M, et al. Interleukin-6 blood level is associated with circulating carcinoembryonic antigen and prognosis in patients with colorectal cancer. Ann Surg Oncol. 2000;7(2):133-138.
[5]Wei LH, Kuo ML, Chen CA, et al. Interleukin-6 promotes cervical tumor growth by VEGF-dependent angiogenesis via a STAT3 pathway. Oncogene. 2003;22(10):1517-1527.
[6]Shinriki S, Jono H, Ota K, et al. Humanized anti-interleukin-6 receptor antibody suppresses tumor angiogenesis and in vivo growth of human oral squamous cell carcinoma. Clin Cancer Res. 2009;15(17):5426-5434.
[7]Duan Z, Lamendola DE, Penson RT, Kronish KM, Seiden MV. Overexpression of IL-6 but not IL-8 increases paclitaxel resistance of U-2OS human osteosarcoma cells. Cytokine. 2002;17(5):234-242.
[8]Liu H, Yan R, Xiao Z, et al. Targeting DCLK1 attenuates tumor stemness and evokes antitumor immunity in triple-negative breast cancer by inhibiting IL-6/STAT3 signaling. Breast Cancer Res. 2023;25(1):43.
[9]Song L, Rawal B, Nemeth JA, Haura EB. JAK1 activates STAT3 activity in non-small-cell lung cancer cells and IL-6 neutralizing antibodies can suppress JAK1-STAT3 signaling. Mol Cancer Ther. 2011;10(3):481-494.
[10]Thilakasiri P, Huynh J, Poh AR, et al. Repurposing the selective estrogen receptor modulator bazedoxifene to suppress gastrointestinal cancer growth. EMBO Mol Med. 2019;11(4):e9539.
[11]Wolf CL, Feci A, Tuccinardi JP, et al. Development of the First Small-Molecule Inhibitor Targeting Oncostatin M for Treatment of Breast Cancer. J Med Chem. 2025;68(15):15422-15445.
[12]Shi Y, Gao W, Lytle NK, et al. Targeting LIF-mediated paracrine interaction for pancreatic cancer therapy and monitoring. Nature. 2019;569(7754):131-135.
[13]Liang B, Gardner DB, Griswold DE, Bugelski PJ, Song XY. Anti-interleukin-6 monoclonal antibody inhibits autoimmune responses in a murine model of systemic lupus erythematosus. Immunology. 2006;119(3):296-305.
[14]Kitaba S, Murota H, Terao M, et al. Blockade of interleukin-6 receptor alleviates disease in mouse model of scleroderma. Am J Pathol. 2012;180(1):165-176.
[15]Elshabrawy HA, Volin MV, Essani AB, et al. IL-11 facilitates a novel connection between RA joint fibroblasts and endothelial cells. Angiogenesis. 2018;21(2):215-228.
[16]Liu Q, Du Y, Li K, et al. Anti-OSM Antibody Inhibits Tubulointerstitial Lesion in a Murine Model of Lupus Nephritis. Mediators Inflamm. 2017;2017:3038514.
[17]Okuno S, Hashimoto T, Yamazaki Y, Okuzawa M, Satoh T. IL-31 and IL-31 receptor alpha in pemphigus: Contributors to more than just itch?. J Dermatol. 2023;50(7):927-930.
[18]Fanola CL, Morrow DA, Cannon CP, et al. Interleukin-6 and the Risk of Adverse Outcomes in Patients After an Acute Coronary Syndrome: Observations From the SOLID-TIMI 52 (Stabilization of Plaque Using Darapladib-Thrombolysis in Myocardial Infarction 52) Trial. J Am Heart Assoc. 2017;6(10):e005637.
[19]Held C, White HD, Stewart RAH, et al. Inflammatory Biomarkers Interleukin-6 and C-Reactive Protein and Outcomes in Stable Coronary Heart Disease: Experiences From the STABILITY (Stabilization of Atherosclerotic Plaque by Initiation of Darapladib Therapy) Trial. J Am Heart Assoc. 2017;6(10):e005077.
[20]Li H, Liu W, Xie J. Circulating interleukin-6 levels and cardiovascular and all-cause mortality in the elderly population: A meta-analysis. Arch Gerontol Geriatr. 2017;73:257-262.
[21]Kobara M, Noda K, Kitamura M, et al. Antibody against interleukin-6 receptor attenuates left ventricular remodelling after myocardial infarction in mice. Cardiovasc Res. 2010;87(3):424-430.
[22]Ridker PM, Devalaraja M, Baeres FMM, et al. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet. 2021;397(10289):2060-2069.
[23]Miteva K, Baptista D, Montecucco F, et al. Cardiotrophin-1 Deficiency Abrogates Atherosclerosis Progression. Sci Rep. 2020;10(1):5791.
[24]Song K, Wang S, Huang B, Luciano A, Srivastava R, Mani A. Plasma cardiotrophin-1 levels are associated with hypertensive heart disease: a meta-analysis. J Clin Hypertens (Greenwich). 2014;16(9):686-692.
[25]Lim WW, Dong J, Ng B, et al. Inhibition of IL11 Signaling Reduces Aortic Pathology in Murine Marfan Syndrome. Circ Res. 2022;130(5):728-740.
[26]Zhang M, Wang C, Hu J, et al. Notch3/Akt signaling contributes to OSM-induced protection against cardiac ischemia/reperfusion injury. Apoptosis. 2015;20(9):1150-1163.
[27]Rolfe BE, Stamatiou S, World CJ, et al. Leukaemia inhibitory factor retards the progression of atherosclerosis. Cardiovasc Res. 2003;58(1):222-230.
[28]González GE, Rhaleb NE, D'Ambrosio MA, et al. Deletion of interleukin-6 prevents cardiac inflammation, fibrosis and dysfunction without affecting blood pressure in angiotensin II-high salt-induced hypertension. J Hypertens. 2015;33(1):144-152.
[29]Chen W, Yuan H, Cao W, et al. Blocking interleukin-6 trans-signaling protects against renal fibrosis by suppressing STAT3 activation. Theranostics. 2019;9(14):3980-3991.
[30]Wheeler DS, Misumi K, Walker NM, et al. Interleukin 6 trans-signaling is a critical driver of lung allograft fibrosis. Am J Transplant. 2021;21(7):2360-2371.
[31]Schafer S, Viswanathan S, Widjaja AA, et al. IL-11 is a crucial determinant of cardiovascular fibrosis. Nature. 2017;552(7683):110-115.
[32]Ng B, Dong J, D'Agostino G, et al. Interleukin-11 is a therapeutic target in idiopathic pulmonary fibrosis. Sci Transl Med. 2019;11(511):eaaw1237.
[33]Matsuda M, Tsurusaki S, Miyata N, et al. Oncostatin M causes liver fibrosis by regulating cooperation between hepatic stellate cells and macrophages in mice. Hepatology. 2018;67(1):296-312.
[34]Xu S, Yang X, Chen Q, et al. Leukemia inhibitory factor is a therapeutic target for renal interstitial fibrosis. EBioMedicine. 2022;86:104312.
[35]Perretta-Tejedor N, Muñoz-Félix JM, Düwel A, et al. Cardiotrophin-1 opposes renal fibrosis in mice: Potential prevention of chronic kidney disease. Acta Physiol (Oxf). 2019;226(2):e13247.
