Rat Model for Sciatic Nerve Injury (SNI)
Nervi ischiadicus
- Product No.DSI838Ra01
- Organism SpeciesRattus norvegicus (Rat) Same name, Different species.
- Prototype SpeciesHuman
- Sourcecaused by clamping on sciatic nerve trunk
- Model Animal StrainsSPF SD rat, male, age:8 weeks
- Modeling GroupingRandomly divided into groups: Control group, Model group, Positive drug group and Test drug group, 15 mice per group
- Modeling Period4 weeks
- Modeling MethodModeling : Firstly,the male Wistar rats were injected with 10% chloral hydrate according to the dose of 3.5ml/kg by intraperitoneal injection. Fixed it on the hohder in a prone position, spreaded a towel, exposed left lower limbs and cut off hair. Then used iodine and alcohol to disinfect for three times, cut skin for 6-8mm at median line of the left femur to expose muscle. Secondary, separated biceps femoris, semitendinosus and semi-membrane muscle by a haemostatic forceps to isolate the white and thick sciatic nerve which was stimulated to cause a left foot twitching. At 6-8mm from the ischial tuberosity, closed ischiadic nerve by a hemostatic forceps for three pressures. After 5 seconds of pressure , release the hemostatic forceps for 10 seconds. The width of the crush injury was about 3mm. Finally, after marking 9-0 noninvasive suture, the sciatic nerve was returned back in situ, the muscle was arranged and the wound was sutured. Rats in the sham operation group, after narcotized, only cut the skin to expose left sciatic nerve without pressure, and the corresponding positions were marked and sutured with the same method.
Sample collection:
After feeding the animals for a week, the rats were injected with chloral hydrate and fixed on the hohder in a supine position. From the lower jaw to the lower abdomen, and the skin was cut through a combined incision. Cut the ribs and diaphragm to expose the heart. Punctured into left ventricle from the left ventricle with a 16 needle and inserted the aorta along the left ventricle into the aorta with the soft indwelling catheter. And the right atrial appendage was cut. Firstly, washed blood in the tissue with 250ml 0.9% normal saline, then rapidly added 100ml 4% Polyformaldehyde stationary solution, then added 100ml slowly Until the tail was raised, the body was harden and the color turned white. Immediately fixed rats whose body was harden on the operating table in the prone position, then cut skin along the spine from chest to sacrum to expose and separate dorsal muscle. Secondary, cut off spine and spinal cord of T12 L1 level along 12 costal muscles, Along the sciatic nerve, cut the sciatic nerve of waist 4, waist 5. sacrum 1, including the sciatic nerve within 2mm crush injury. The corresponding positions were cut in the sham operation group with the same method. The three methods for processing and fixing: (1) fixed with 4% paraformaldehyde stationary solution to prepare parafin section. (2) fixed it with 2.5% glutaraldehyde solution for electron microscope (3) freezed in liquid nitrogen to prepare frozen slices. - ApplicationsDisease Model
- Downloadn/a
- UOM Each case
- FOB
US$ 240
For more details, please contact local distributors!
Model Evaluation
Pathological Results
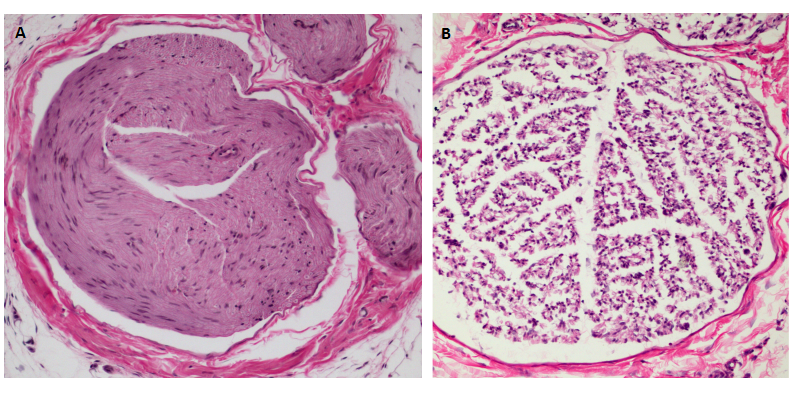
1. Pathological examination of rats sciatic tissue
The sciatic nerve tissue was fixed in 4% paraformaldehyde for 24 hours, then embedded into paraffin. The paraffin block was frozen at -20 ℃ and sliced for a thickness of 4um. Unfold the slice in the water, then roasted it at 60 ℃ for 2 hours. Stained it with Loyez hematoxylin, and observed the condition of myelin sheath under light microscope.
2. Sciatic nerve tissue apoptosis by TUNEL detection
The slices of sciatic nerve were dewaxed,and digested by protease K at room temperature, and incubated with TDT enzyme for 1 hour, and 0.3%H2O2 inhibited endogenous peroxidase. Streptavidin-HRP at room temperature for 5 min. Colorated it with DBA and restained it with hematoxylin. The negative control group used enzyme free labeled solution to instead TDT enzyme. The apoptotic nucleus was brown granules and the apoptotic cells were observed and counted under light microscope.
Cytokines Level
Statistical Analysis
SPSS software is used for statistical analysis, measurement data to mean ± standard deviation (x ±s), using t test and single factor analysis of variance for group comparison, P<0.05 indicates there was a significant difference, P<0.01 indicates there are very significant differences.
GIVEAWAYS
INCREMENT SERVICES
-
 Tissue/Sections Customized Service
Tissue/Sections Customized Service
-
 Serums Customized Service
Serums Customized Service
-
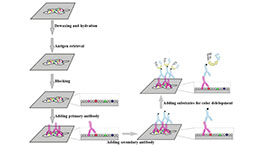 Immunohistochemistry (IHC) Experiment Service
Immunohistochemistry (IHC) Experiment Service
-
 Small Animal In Vivo Imaging Experiment Service
Small Animal In Vivo Imaging Experiment Service
-
 Small Animal Micro CT Imaging Experiment Service
Small Animal Micro CT Imaging Experiment Service
-
 Small Animal MRI Imaging Experiment Service
Small Animal MRI Imaging Experiment Service
-
 Small Animal Ultrasound Imaging Experiment Service
Small Animal Ultrasound Imaging Experiment Service
-
 Transmission Electron Microscopy (TEM) Experiment Service
Transmission Electron Microscopy (TEM) Experiment Service
-
 Scanning Electron Microscope (SEM) Experiment Service
Scanning Electron Microscope (SEM) Experiment Service
-
 Learning and Memory Behavioral Experiment Service
Learning and Memory Behavioral Experiment Service
-
 Anxiety and Depression Behavioral Experiment Service
Anxiety and Depression Behavioral Experiment Service
-
 Drug Addiction Behavioral Experiment Service
Drug Addiction Behavioral Experiment Service
-
 Pain Behavioral Experiment Service
Pain Behavioral Experiment Service
-
 Neuropsychiatric Disorder Behavioral Experiment Service
Neuropsychiatric Disorder Behavioral Experiment Service
-
 Fatigue Behavioral Experiment Service
Fatigue Behavioral Experiment Service
-
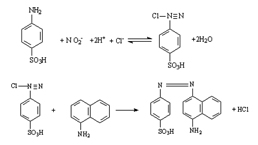 Nitric Oxide Assay Kit (A012)
Nitric Oxide Assay Kit (A012)
-
 Nitric Oxide Assay Kit (A013-2)
Nitric Oxide Assay Kit (A013-2)
-
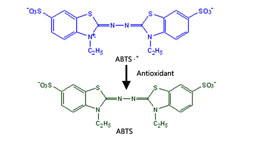 Total Anti-Oxidative Capability Assay Kit(A015-2)
Total Anti-Oxidative Capability Assay Kit(A015-2)
-
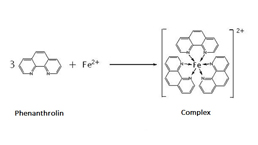 Total Anti-Oxidative Capability Assay Kit (A015-1)
Total Anti-Oxidative Capability Assay Kit (A015-1)
-
 Superoxide Dismutase Assay Kit
Superoxide Dismutase Assay Kit
-
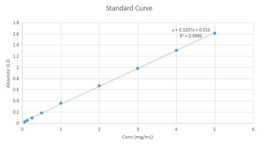 Fructose Assay Kit (A085)
Fructose Assay Kit (A085)
-
 Citric Acid Assay Kit (A128 )
Citric Acid Assay Kit (A128 )
-
 Catalase Assay Kit
Catalase Assay Kit
-
 Malondialdehyde Assay Kit
Malondialdehyde Assay Kit
-
 Glutathione S-Transferase Assay Kit
Glutathione S-Transferase Assay Kit
-
 Microscale Reduced Glutathione assay kit
Microscale Reduced Glutathione assay kit
-
 Glutathione Reductase Activity Coefficient Assay Kit
Glutathione Reductase Activity Coefficient Assay Kit
-
 Angiotensin Converting Enzyme Kit
Angiotensin Converting Enzyme Kit
-
 Glutathione Peroxidase (GSH-PX) Assay Kit
Glutathione Peroxidase (GSH-PX) Assay Kit
-
 Cloud-Clone Multiplex assay kits
Cloud-Clone Multiplex assay kits
| Catalog No. | Related products for research use of Rattus norvegicus (Rat) Organism species | Applications (RESEARCH USE ONLY!) |
| DSI838Ra01 | Rat Model for Sciatic Nerve Injury (SNI) | Disease Model |
| TSI838Ra79 | Rat Sciatic nerve Tissue of Sciatic Nerve Injury (SNI) | Paraffin slides for pathologic research: IHC,IF and HE,Masson and other stainings |